About Celltech
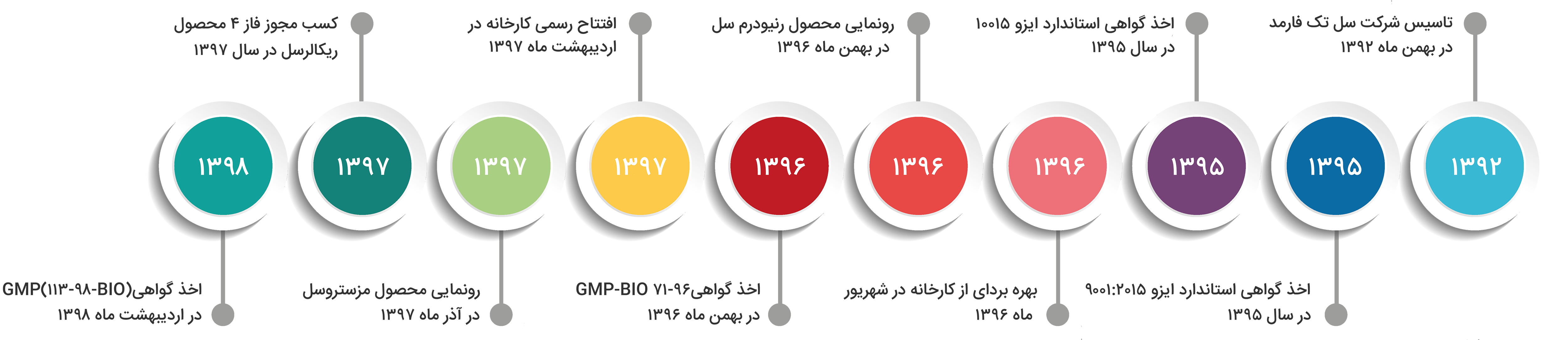
Celltech Pharmed-Stem Cell company was established in February 2013. In 2016, it succeeded in obtaining the ISO 9001: 2015 standard certificate and the ISO 10015 standard certificate. In September 2017, Celltech factory was put into operation. The Renodermcell product was unveiled in February 2017 by this company and following that GMP certificate (BIO-96-71) was obtained. The factory was officially opened in May 2016. In December 2016, the product of Mesestrocell was unveiled, and the license for phase 4 of Recallcell product was obtained. In May 2009, Celltech Pharmed-Stem Cell received the GMP certificate (BIO-98-113), and in June 2009, it was able to renew the ISO 9001: 2015 standard certificate.
۲۰۱۳: Establishment of Celltech Pharmed-Stem Cell company in February 2013
۲۰۱۶: Obtaining ISO 9001: 2015 standard certificate in 2016
۲۰۱۶: Obtaining ISO 10015 standard certificate in 2016
۲۰۱۷: Operation of the factory in September 2017
۲۰۱۷: Showcase of Renodermcell product in February 2017
۲۰۱۷: Obtaining GMP-BIO 71-96 certificate in February 2017
۲۰۱۸: Official opening of the factory in May 2018
۲۰۱۸: The unveiling of the Mesestrocell product in December 2018
۲۰۱۸: Obtaining Phase 4 license for Recallcell product in 2018
۲۰۱۹: Obtaining a GMP (BIO-113-98) certificate in May 2019